Company Statement
Clarification to article: “Novel vaccine technologies impact human and salmon health outcomes” (Fish Farmer Yearbook, 2022).
Regarding the article published in the 2022 edition of the Fish Farmer Yearbook entitled ‘Novel vaccine technologies impact human and salmon health outcomes’, and in response to a complaint, Elanco would like to clarify the following points:
1. In describing the advantages of DNA vaccines over conventional vaccines it was implied that DNA vaccines hold an advantage regarding the plasmid stability and hence a longer product shelf life facilitating ease of storage and shipping. As approximately 50% of the approved PD vaccines in the UK for use in Atlantic Salmon offer two years of shelf life, the same shelf life as CLYNAV™, we acknowledge that our statement cannot be applied generally.
2. The statement “…These vaccines are extremely safe because they: cannot cause infection (they don’t contain a micro-organism or the genome for a complete organism)…” could, if taken out of context, imply that conventional PD vaccines in the market are less safe. This statement is correct with regard to the mode of action of live attenuated vaccines. These vaccines require the infection of the host in order to generate a protective immune response. It is generally accepted that attenuated vaccines produce transient side effects sometimes even consistent with the pathology they are intended to prevent (see footnotes 1, 2). It should be made clear that all current conventional PD vaccines used within the UK market are based on inactivated viral vaccine technology. All medicines, including vaccines, which have a Marketing Authorization in the UK have undergone rigorous risk benefit evaluation and are deemed to be safe and effective.
The use of DNA plasmid vaccine technology does, however, offer distinct advantages when compared with a conventional oil adjuvated inactivated viral PD vaccine. The development of technology which precludes the use of mineral oil has significant impacts on the reduction of risk for the fish (see footnotes 3, 4). The use of mineral oil adjuvants in ‘traditional’ vaccines can lead to variable degrees of side effects in the form of melanisation and adhesions (see footnote 5). In order to qualify this point, reference is made to Table 1., which states the adverse reactions registered in the UK approved SPC* for both CLYNAV™ and ALPHA JECT® micro 1 PD. Oil-adjuvanted vaccines have been mentioned in the recently reported ‘cross-stitch’ pathology identified in Norway (see footnote 6).
As previously mentioned, the use of oil-adjuvated vaccines can cause variable levels of adhesions and melanisation in the peritoneal cavity (see footnote 5).
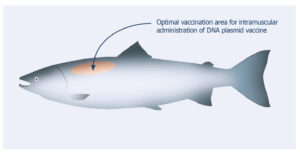
These side effects are well documented (see footnotes 5, 7, 8, 9, 10). The fact that CLYNAV™ is administered by intramuscular injection in the area immediately anterior and lateral to the dorsal fin, in the epaxial muscle, and does not contain a mineral oil adjuvant mitigates against these documented reactions (see footnote 11).
Getting things wrong when vaccinating via the intraperitoneal route using oil-adjuvated vaccines can negatively affect fish welfare, growth, and result in costly downgrades at harvest due to the described side effects of melanisation, adhesions, and spinal deformity (see footnotes 6, 10).

Figure 1. Location of CLYNAV™ vaccine administration

Table 1. Key differences in the SPC* of CLYNAV™ and ALPHA JECT® micro 1PD (see footnotes 11, 12)
† The frequency of adverse reactions is defined using the following convention:
• Very common (more than 1 in 10 animals treated displaying adverse reaction(s))
• Common (more than 1 but less than 10 animals in 100 animals treated)
• Uncommon (more than 1 but less than 10 animals in 1,000 animals treated)
• Rare (more than 1 but less than 10 animals in 10,000 animals treated)
• Very rare (less than 1 animal in 10,000 animals treated, including isolated reports).
CLYNAV™: A new era in vaccine technology
Novel vaccine mode of action
CLYNAV™’s DNA plasmid instructs cells adjacent to the site of injection to produce specific antigens of interest for salmon alpha virus (SAV) which can then be presented to the immune system. CLYNAV™’s DNA plasmid is taken up by the cells adjacent to the site of injection and directs production of the antigens using the host cell machinery. These antigens are recognized as foreign proteins and are then processed and displayed on the cell surface triggering a broad, potent immune response including both antibody (humoral) and cell-mediated components. Figure 2 illustrates how CLYNAV™ uses the myocytes and the antigen presenting cells (APC) to trigger the cell-mediated (CD8* T cells with MHC I) and humoral immunity pathways (CD4* T cells with MHC II), respectively (see footnotes 4, 13).

Figure 2. How CLYNAV™ produces humoral and cell-mediated immunity
Proven high performance PD control
A study was conducted independently, measuring the efficacy of PD vaccines in commercially reared Atlantic salmon, using mortality and growth as the key outcome variables. This study was conducted on commercial farming sites where the presence of either, or both, SAV2 and SAV3 subtypes were detected (see footnote 14).
The below vaccine groups were assessed:

The cumulative mortality until harvest for the salmon vaccinated with CLYNAV™ was the lowest (2.9%) compared to an overall cumulative mortality of 4.2% for all the groups combined. The cumulative mortality for the group vaccinated with ALPHA JECT® micro 1PD was 3.7% (see footnote 14).
The CLYNAV™ vaccinated group also had higher slaughter weights at harvest than all the other groups including fish vaccinated with ALPHA JECT® micro 1PD. In fact the CLYNAV™ vaccinated fish were 0.67kg heavier than those vaccinated with ALPHA JECT® micro 1PD (refer Figure 3) (see footnote 14).

Figure 3. Average weight of different study groups at slaughter (kg) (see footnote 14).

References: 1. Mohd-Aris, Aslizah; Muhamad-Sofie, Mohd Hafiz Ngoo; Zamri-Saad, Mohd; Daud, Hassan Mohd; Ina-Salwany, Md. Yasin. Live vaccines against bacterial fish diseases: A review. 2019. Veterinary World Publisher; 12 (11): 1806-1815. 2. Minor Philip D. Live attenuated vaccines: Historical successes and current challenges. 2015. Virology; 479-480: 379-392. 3. Lorenzen N, LaPrata SE. DNA vaccines for aquacultured fish. 2005. Re. sci. tech. Off. Int. Epiz; 24 (1): 201-213. 4. C. Collins et al. DNA vaccination for finfish aquaculture. 2019. Fish and Shellfish Immunology; 85: 106-125. 5. Midtlyng PJ, Reitan LJ, Speilberg L. Experimental studies on the efficacy and side effects of intraperitoneal vaccination of Atlantic salmon (Salmo salar L.) against furunculosis. 1996. Fish Shellfish Immunol; 6: 335-350. 6. Letter dated 1st October 2020 from the Norwegian Veterinary Institute (NVI) to the Norwegian Ministry of Trade Industry and Fisheries (NFD) regarding the review commissioned by the NFD: Reply regarding vaccination against PD and fish welfare: https://www.vetinst.no/rapporter-og-publikasjoner/faglige-vurderinger-og-horingssva. 7. Midtlyng PJ, Reitan LJ, Lillehaug A, Ramstad A. Protection, immune responses and side effects in Atlantic salmon (Salmo salar L.) vaccinated against furunculosis by different procedures. 1996. Fish Shellfish Immunol; 6: 599-613. 8. Poppe TT, Breck O. Pathology of Atlantic salmon Salmo salar intraperitoneally immunised with oil adjuvanted vaccine. A case report. 1997. Dis Aquat Org 29: 219-226. 9. Brown LA, Hunter D, Thorarinsson R. Fish Vaccination – Methodologies and outcomes. 2003. Fish Veterinary Journal;7: 56-66. 10. Mutoloki S, Alexandersen S, Evensen Ø. Sequential study of antigen persistence and concomitant inflammatory reactions relative to side-effects and growth of Atlantic salmon (Salmo salar L.) following intraperitoneal injection with oil-adjuvanted vaccines. 2004. Fish & Shellfish Immunology; 16: 633-644. 11. https://www.ema.europa.eu/en/documents/product-information/clynav-epar-product-information_en.pdf; accessed on 01/04/2022. 12. https://www.vmd.defra.gov.uk/productinformationdatabase/files/SPC_Documents/SPC_916517.PDF; accessed on 01/04/2022 13. McDonnell WM, Askari FK. The Emerging Role of DNA Vaccines. MedGenMed 1(3), 1999 [formerly published in Medscape Pulmonary Medicine eJournal 2(4), 1998].
14. Magnus V. Røsæg, Arnfinn Aunsmo, Ragnar Thorarinsson. Effect of vaccines against pancreas disease in commercially reared Atlantic Salmon. 2021. Journal of Fish diseases; 44(12): 1911-1924.
Clynav™ contains pUK-SPDVpoly2#1 DNA plasmid coding for salmon pancreas disease virus proteins: 6.0 – 9.4 μg. Legal category in UK.
For further information contact Elanco Animal Health on +44(0)1256 353131 or write to: Elanco Animal Health, Form 2, Bartley Way, Bartley Wood Business Park, Hook, RG27 9XA, UK. Use medicines responsibly www.noah.co.uk/responsible. Advice should be sought from the Medicine Prescriber. Clynav™, Elanco and the diagonal bar logo are trademarks of Elanco or its affiliates. All other product and company names are trademarks of their respective owners. ©2022 Elanco. PM-UK-22-0249

